A human-derived, xeno-free supplement that supports reliable, heparin-free cell expansion in research, translational and clinical applications.
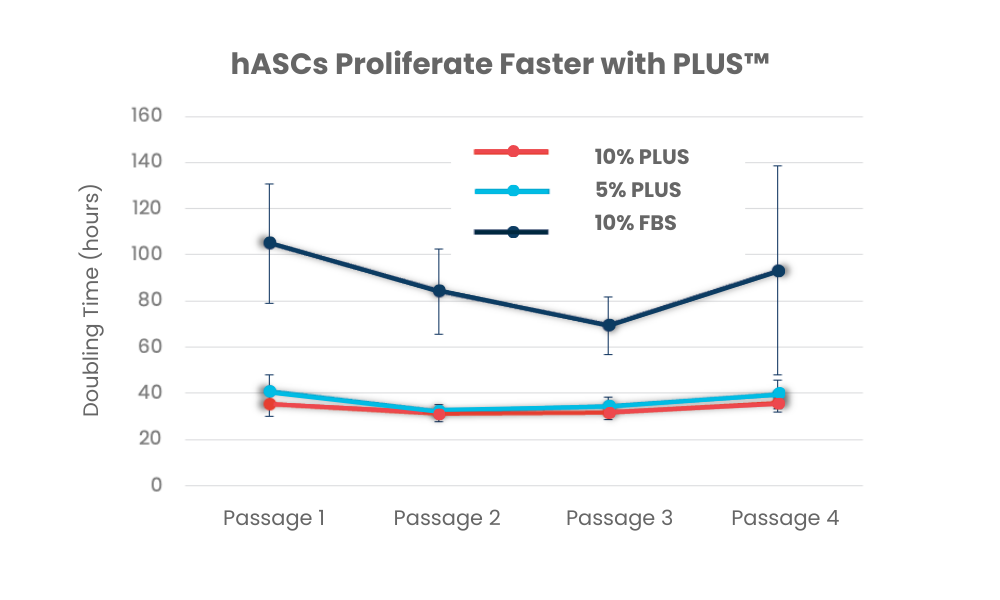
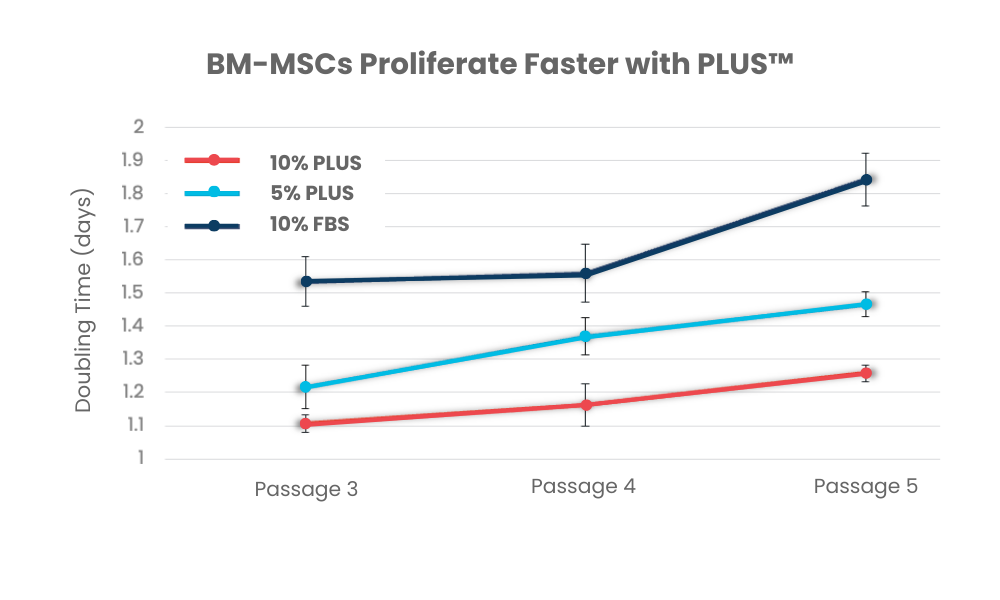
PLUS™ is a pooled human platelet-derived lysate designed for the rapid expansion of human cells in research and clinical applications. PLUS™ is made from transfusion-grade platelets derived from FDA-registered and AABB-accredited blood centers and is a consistent, xeno-free alternative to fetal bovine serum. It is also used as a superior medium compared to human serum for cell culture. It is rich in growth factors and free of animal components and supports the robust proliferation of a wide range of cell types, including MSCs, dendritic cells, fibroblasts, T cells and others.
Descended from humans & effective
Xeno-free & heparin-free
Clinically proven safety
Consistent & scalable
Fibrinogen depleted for all products
Large batch sizes of 100+L
Bags and bottles available
PLUS™ human platelet lysate is available in three flexible grades to support every phase - from early research to clinical application.
All basic functions, plus:
All basic functions, plus:
All basic functions, plus:
Research grade
GMP class
Gamma-irradiated
Price Match Guarantee - We strive to offer the best price. If you find a lower price for a PLUS product, we will match it.