Are you purchasing ancillary materials such as Human Platelet Lysate for your cell therapy manufacturing protocol? Confused by the Clinical grade and GMP grade terminology used by vendors of Ancillary Materials? A recent publication by Solomon et al. in Cytotherapy, titled “Current perspectives on the use of ancillary materials for the manufacture of cellular therapies” addresses the inconsistent use of such terms across the cell therapy industry. Some of the highlights from the publication are discussed below to help researchers who are sourcing hPL cell therapy manufacturing. Ancillary materials (AMs) are components used for manufacturing cell therapy products but are not intended to be part of the final product. Since AMs can significantly impact the efficacy of the final product or the success of a clinical trial or product development, they should be carefully selected and qualified during the process development phase. It is therefore important for AM users to fully understand the claims made by suppliers and hold suppliers accountable for AM labeling and marketing claims. Clinical Grade is used to describe products or materials that are suitable for direct therapeutic use, such as, injectable grade. Such materials are required to show to safety and efficacy for human use through appropriate clinical trials and regulatory approvals. Usually, clinical-grade products are approved as drugs by regulators, and labeling or product documentation should state sterility and safety profile. On the other hand, GMP grade or cGMP grade refers to products manufactured under Current Good Manufacturing Practices which require manufacturers ensure that their products are traceable, safe, pure and effective. This term therefore denotes that a product has been prepared under cGMP guidelines to ensure proper design, monitoring and control of the manufacturing processes, facilities and the final product. Regional differences exist where some national regulatory agencies provide GMP certificates to manufacturers of GMP AMs and others do not. Therefore, when a manufacturer contacts a vendor of a specific AM and comes across the claim “Clinical Grade”, they should ask the vendor for the product’s specific regulatory approvals and safety data. On the other hand, if an AM is labeled as a GMP grade product, the user should qualify the vendor to confirm that the product was manufactured under a quality system compliant to cGMP guidelines. Compass Biomedical offers GMP grade PLUS™ Human Platelet Lysate that is manufactured under a robust quality system. We have been successfully audited by numerous clients who use GMP grade PLUS™ as an ancillary material in their cell therapy protocols. Our quality and regulatory teams are available to assist new clients with quality system audits and obtaining faster approval for clinical trials by using the PLUS™ Drug Master File on record with the US FDA. Reference:
2 Comments
Generation of Monocyte Derived Dendritic Cells Using Xeno-free GMP Grade Human Platelet Lysate3/1/2017 
Human platelet lysate (hPL) is becoming an increasingly popular choice as a xeno-free serum supplement alternative to fetal bovine serum (FBS) for culture expansion of numerous other cell types. Our recent research focuses on the generation of monocyte derived dendritic cells using hPL instead of FBS or human AB serum (hABS).
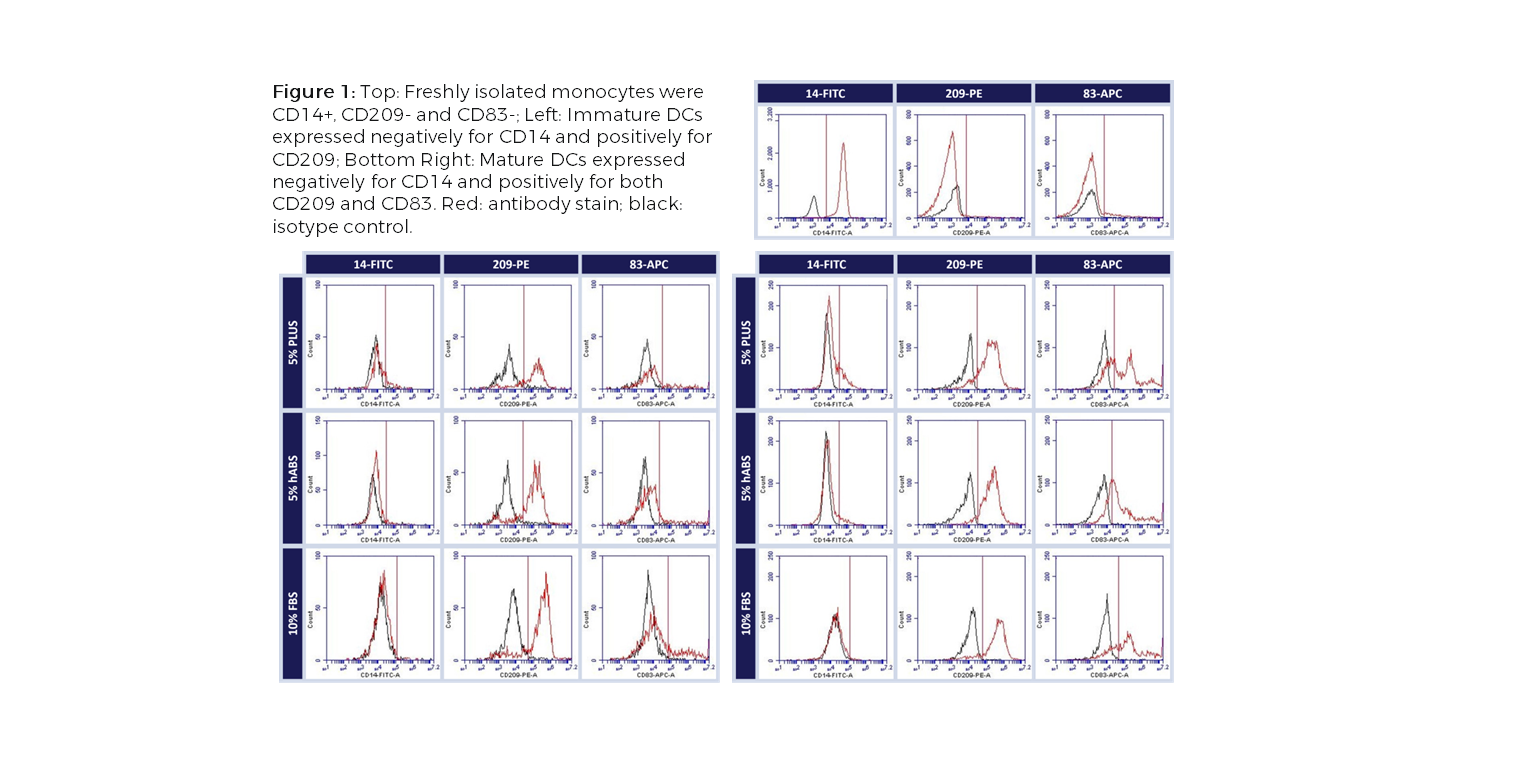
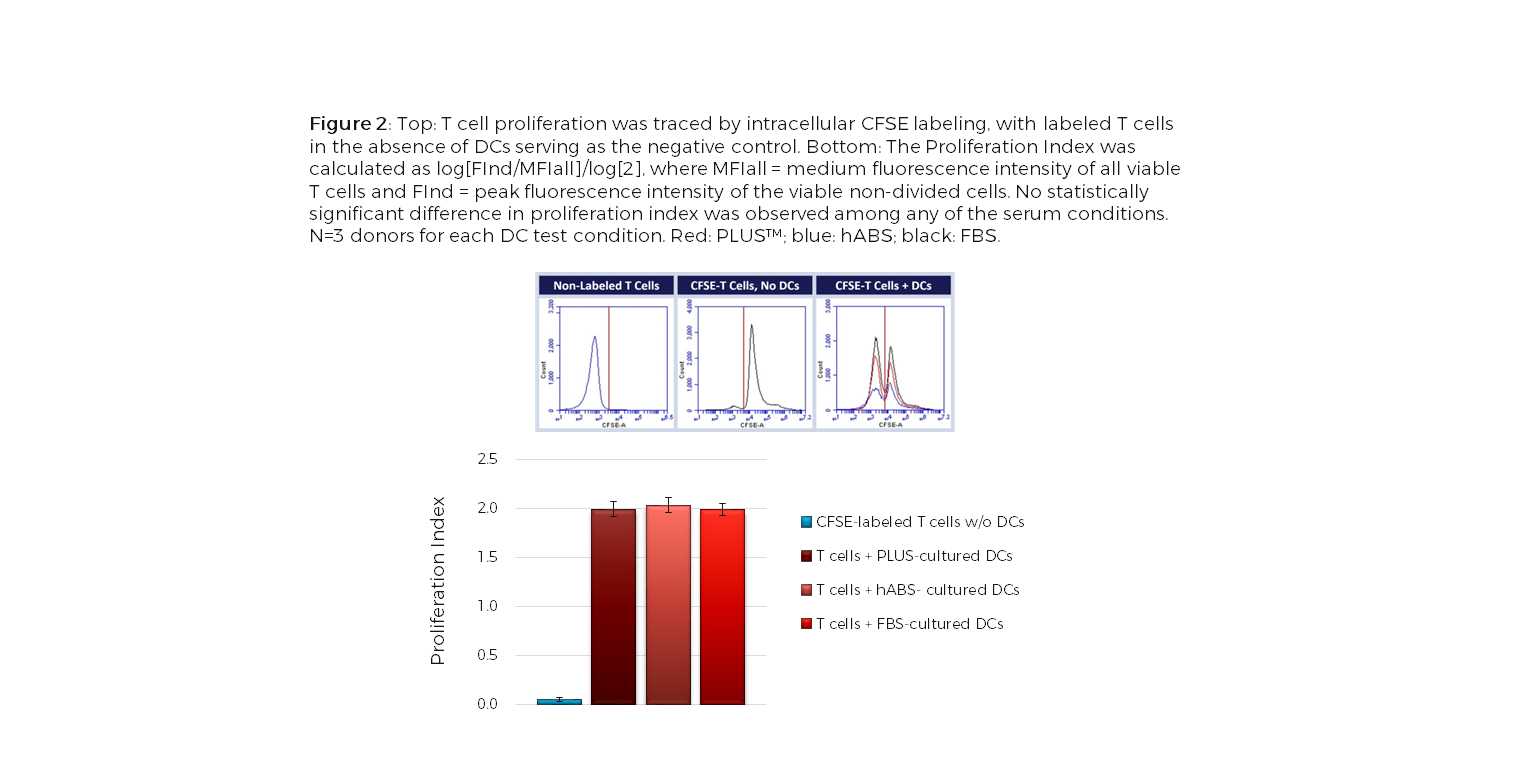
Dendritic cells (DCs), an important antigen-presenting cell type, have been widely used for therapeutic vaccine development in cancer immunotherapy. Among the different approaches to obtain DCs, ex vivo generation from peripheral blood isolated monocytes has been the most favorable method. The ex vivo differentiation of monocytes to immature DCs and their activation to mature DCs require addition of a serum supplement to the culture media. Limitations with Current Choice of Serum: Traditionally, clinical manufacturing of Mo-DCs has employed FBS or human AB serum (hABS). The use of FBS in cell therapy processes poses potential risks for viral and prion transmission as well as adverse immunological reactions while hABS which is collected from a small number of donors has considerable lot-to-lot inconsistency. These variations can significantly impact cell growth, morphology, and functionality, which is of concern with DCs because of their diverse phenotypic range. To address these issues, Compass Biomedical’s PLUS™ human platelet lysate was tested as a replacement for FBS and hABS in culture medium for differentiation and maturation of DCs from peripheral blood isolated monocytes. PLUS™ human platelet lysate is manufactured using a GMP production process with large lots of platelet units obtained from AABB-accredited blood banks, making it a more reliable alternative to hABS and a xeno-free alternative to FBS. Results Flow cytometry analysis of immature and mature Mo-DCs cultured in either PLUS™, hABS or FBS showed that PLUS™ produced mature DCs that were functionally equivalent to those cultured in FBS and hABS (CD14-, CD209+, CD83+, CD86+ and HLA-DR+), independent of the maturation pathway (TNF-α, IFN-γ or LPS) (Figure 1). A functional co-culture assay using PLUS™-cultured mature DCs showed that they stimulated allogeneic T cell proliferation in vitro as efficiently as Mo-DCs cultured in other serum (Figure2). Together, the results show that PLUS™ human platelet lysate can successfully replace FBS or hABS for large-scale ex vivo production of clinical grade Mo-DCs for immunotherapy trials. For further experimental details and results, check out our poster presented at the 2016 ISCT North America conference. |
|
Compass Biomedical 45 South Street Hopkinton, MA 01748, USA Email: [email protected] Phone: 508-283-2622 |
Copyright© 2020 Compass Biomedical, Inc. All rights reserved.